
As a consequence, the consumption of alternatives to animal products increased (BMEL 2021). For instance, the percentage of vegetarians and vegans in Germany doubled from 5 to 10% between 20. 
According to an Australian survey plant-based diets gained in popularity and the number of vegetarians or vegans in the population increased (Roy Morgan 2016). In the last decade, dietary habits have changed in western countries. Especially lower concentrations of ZEN, α-ZEL and α-ZAL (0.001-0.01 nM) in combination with low concentrations of GEN, DAI and EQ (0.001-0.1 µM) strongly increased the estrogenic response compared to the single substances. Most combinations of isoflavones with mycoestrogens enhanced the estrogenic response in the investigated concentrations. For the individual substances the following order of estrogenicity was obtained: α-ZEL > α-ZAL > ZEN > GEN > EQ > DAI > GLY. In the case of mycoestrogens, the tested concentration range included 0.001 to 10 nM with multiplication steps of 10 in between, while for the isoflavones 1000 times higher concentrations were investigated. In the present study, we investigated the estrogenic potential of mycoestrogens and the isoflavones genistein (GEN), daidzein (DAI) and glycitein (GLY) as well as equol (EQ), the gut microbial metabolite of DAI, in vitro alone or in combination, using the alkaline phosphatase (ALP) assay in Ishikawa cells. So far, risk assessment of potential endocrine disruptors is usually based on adverse effects of single compounds whereas studies investigating combinatorial effects are scarce. In a mixed diet or especially animal feed, these potential contaminants might be ingested together with naturally occurring phytoestrogens such as soy isoflavones. 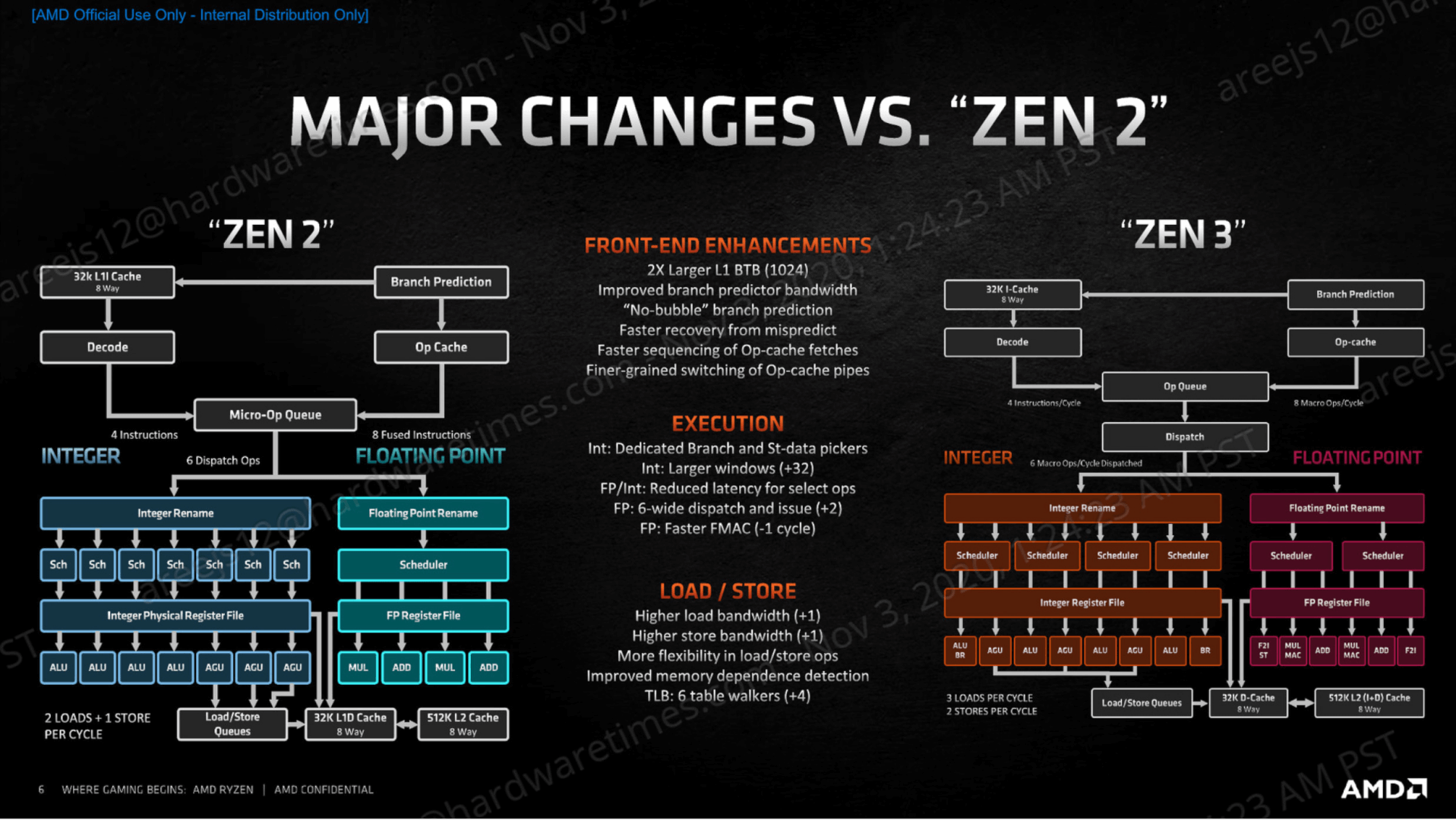
Among these substances, the Fusarium mycoestrogen zearalenone (ZEN) and its metabolites α-zearalenol (α-ZEL) and α-zearalanol (α-ZAL) are known to possess endocrine disruptive properties. If the colour of the solution (which is originally orange) turns to green, then the gas is \(SO_2\).Ĭhromium in its +6 oxidation state and in the dichromate (\(Cr_2O_7^\) is green coloured.Humans and animals are exposed to multiple substances in their food and feed that might have a negative health impact. The gas is passed into to an aqueous solution of acidified Potassium Dichromate. Here is the reaction to distinguish between them. 
Visually, the same visual thing is observed when \(CO_2\) and \(SO_2\) is passed through lime water. What is the test to distinguish between the two, then? \(Ca(HSO_3)_2\) is soluble in water and is responsible in making the solution clear again. \(CaSO_3\) is a precipitate and is responsible for the turbidity of the solution. \(SO_2\) when passed through lime water makes it turbid (milky) and if the passage of \(SO_2\) is prolonged the turbidity disappears and the solution becomes clear again. \(Ca(HCO_3)_2\) is soluble in water and is responsible in making the solution clear again. \(CaCO_3\) is a precipitate and is responsible for the turbidity of the solution. \(CO_2\) when passed through lime water makes it turbid (milky) and if the passage of \(CO_2\) is prolonged the turbidity disappears and the solution becomes clear again. These gases are then identified with appropriate tests. For example, carbonate salts will release \(CO_2\) and bisulphite salts release \(SO_2\). While performing Salt Analysis, the action of heat on a salt with or without an acid will release gases that can be used to identify the anion of the salt.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
